 Enzymes work best within specific temperature and pH ranges, and sub-optimal conditions can cause an enzyme to lose its ability to bind to a substrate. Thus, when adding either substrate or enzyme, you should expect to see the see the reaction rate curve level off when the maximum is reached. Enzyme activity can be affected by a variety of factors, such as temperature, pH, and concentration. Add more substrate and there won't be enzyme free to work with it. Add more enzyme, and there won't be substrate for it to react with. Increasing substrate concentration should still increase the reaction rate, but because enzymes can be inhibited regardless of how saturated their active sites.  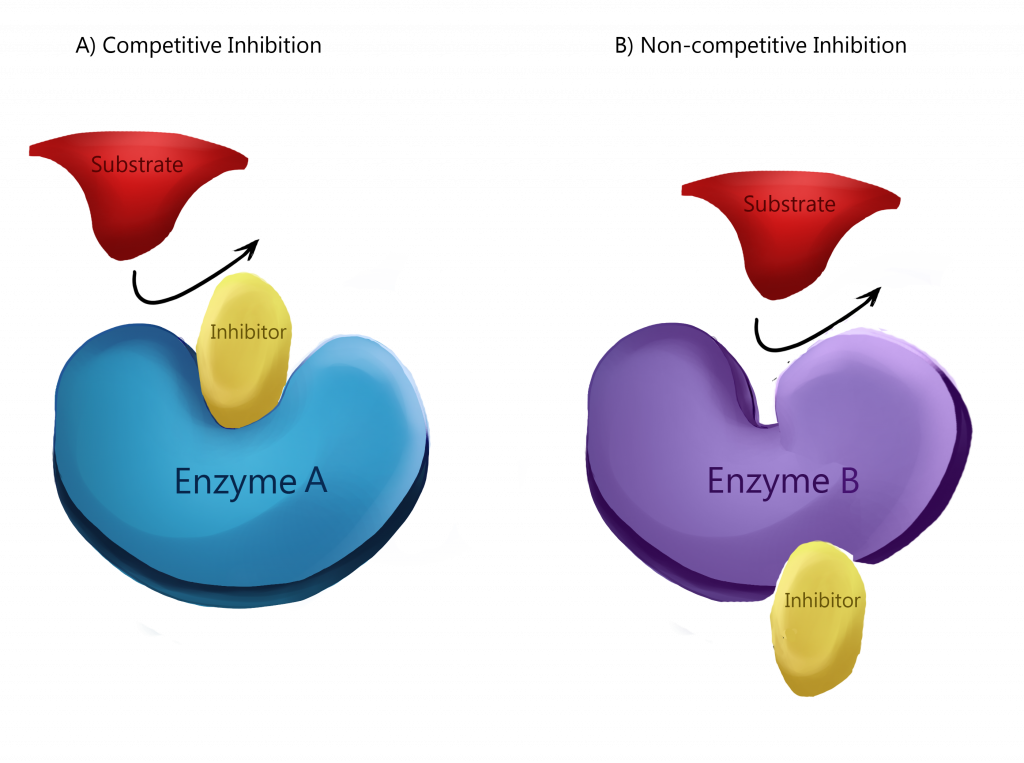
When there is a 1:1 ratio of enzyme and substrate, the reaction is going as fast as it can. At some point, the maximum raction rate should be achieved and we should see the curve level off (see the substrate concentration curve above for an example), but based on this data, more than 3 mL of turnip extract is required to reach a 1:1 ratio of enzyme to substrate molecules.Įnzyme reaction rates have set limits: they can't drop past zero (no reaction) and they can't speed up past 100%. This is represented graphically in Figure 8. As soon as the catalytic site is empty, more substrate is available to bind and undergo reaction. After this point, increases in substrate concentration will not increase the velocity (delta A/delta T). The graph above shows that as we increased the amount of enzyme in the tubes, our reaction rate increased. of substrate increases, the enzyme becomes saturated with substrate. Enzyme promiscuity can confer a fitness benefit in novel growth. When an enzyme binds to its substrate, we know it lowers the activation energy of the reaction, allowing it to happen more quickly. 
S is plotted (Figure 4.35 red line), as well as 1/V0 vs. Since the turnip extract contained our peroxidase enzyme, this increased the enzyme concentration in our reactions. Structural mutations shifted enzyme substrate turnover rates toward the new substrate. S reactions without inhibitor (20 or so tubes, with buffer and constant amounts of enzyme, varying amounts of substrate, equal reaction times). We also examined reaction rate when we increased the amount of turnip extract in our tubes. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
